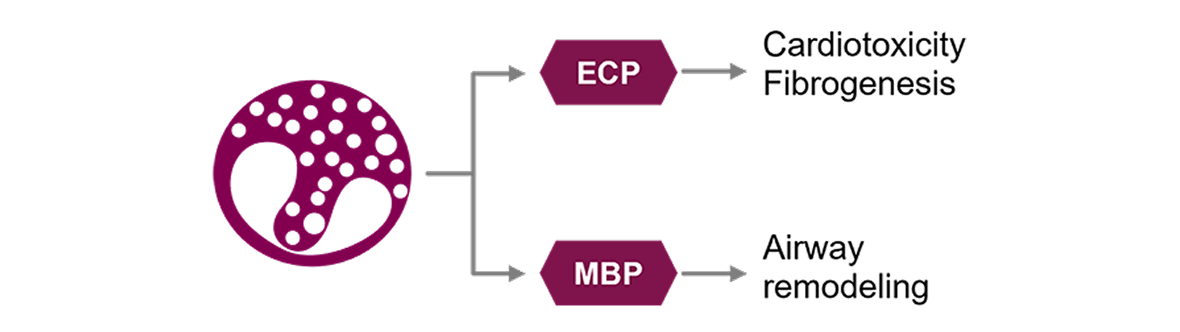
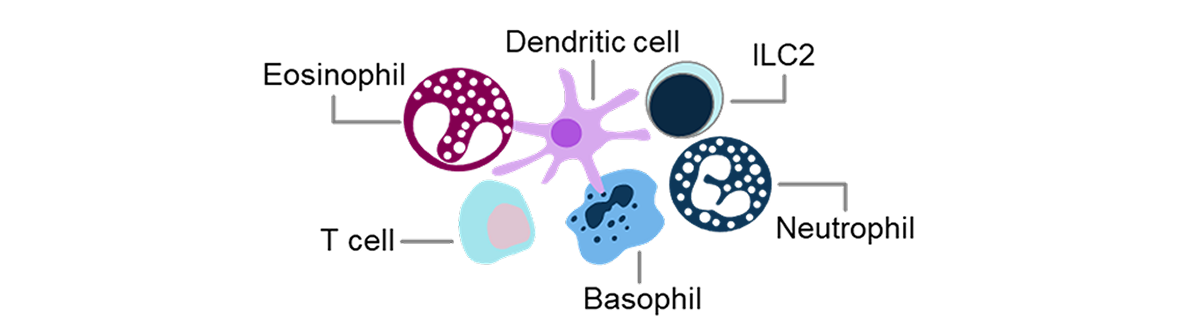
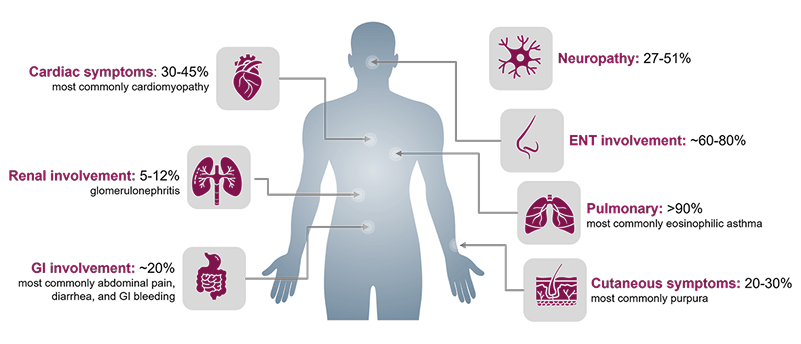
1. Jackson DJ, Akuthota P, Roufosse F. Eosinophils and eosinophilic immune dysfunction in health and disease. Eur Respir Rev. 2022;31(163):210150. doi:10.1183/16000617.0150-2021
2. Jakes RW, Kwon N, Nordstrom B, et al. Burden of illness associated with eosinophilic granulomatosis with polyangiitis: a systematic literature review and meta-analysis. Clin Rheumatol. 2021;40(12):4829-4836. doi:10.1007/s10067-021-05783-8
3. Chung SA, Langford CA, Maz M, et al. 2021 American College of Rheumatology/Vasculitis Foundation guideline for the management of antineutrophil cytoplasmic antibody-associated vasculitis. Arthritis Rheumatol. 2021;73(8):1366-1383. doi:10.1002/art.41773
4. Hellmich B, Sanchez-Alamo B, Schirmer JH, et al. EULAR recommendations for the management of ANCA-associated vasculitis: 2022 update. Ann Rheum Dis. 2024;83(1):30-47. doi:10.1136/ard-2022-223764
5. Bloom JL, Langford CA, Wechsler ME. Therapeutic advances in eosinophilic granulomatosis with polyangiitis. Rheum Dis Clin North Am. 2023;49(3):563-584. doi:10.1016/j.rdc.2023.03.006
6. Yates M, Watts R. ANCA-associated vasculitis. Clin Med (Lond). 2017;17(1):60-64. doi:10.7861/clinmedicine.17-1-60
7. Jennette JC, Falk RJ, Bacon PA, et al. 2012 revised International Chapel Hill Consensus Conference nomenclature of vasculitides. Arthritis Rheum. 2013;65(1):1-11. doi:10.1002/art.37715
8. Fagni F, Bello F, Emmi G. Eosinophilic granulomatosis with polyangiitis: dissecting the pathophysiology. Front Med (Lausanne). 2021;8:627776. doi:10.3389/fmed.2021.627776
9. Sokołowska B, Szczeklik W, Piłat O, et al. The impact of current health-related quality of life on future health outlook in patients with eosinophilic granulomatosis with polyangiitis (Churg-Strauss syndrome). Clin Rheumatol. 2013;32(6):779-785. doi:10.1007/s10067-013-2169-7
10. Benarous L, Terrier B, Laborde-Casterot H, et al. Employment, work disability and quality of life in patients with ANCA-associated vasculitides. The EXPOVAS study. Clin Exp Rheumatol. 2017;35(suppl 103):40-46.
11. Ribi C, Cohen P, Pagnoux C, et al. Treatment of Churg-Strauss syndrome without poor-prognosis factors: a multicenter, prospective, randomized, open-label study of seventy-two patients. Arthritis Rheum. 2008;58(2):586-594. doi:10.1002/art.23198
12. Yates M, Watts RA, Bajema IM, et al. EULAR/ERA-EDTA recommendations for the management of ANCA-associated vasculitis. Ann Rheum Dis. 2016;75(9):1583-1594. doi:10.1136/annrheumdis-2016-209133
13. Chen LF, Qianhua LI, Zheng DH, et al. Comparison of clinical characteristics of eosinophilic granulomatosis with polyangiitis between rheumatology and respiratory medicine: a single center, retrospective study [abstract]. Ann Rheum Dis. 2019;78(suppl 2):1751-1752. Abs AB0579. doi:10.1136/annrheumdis-2019-eular.5369
14. Emmi G, Bettiol A, Gelain E, et al. Evidence-based guideline for the diagnosis and management of eosinophilic granulomatosis with polyangiitis. Nat Rev Rheumatol. 2023;19(6):378-393. doi:10.1038/s41584-023-00958-w
15. Vasculitis Foundation. Pediatric vasculitis. Who gets EGPA? Vasculitis Foundation website. Accessed February 11, 2026. https://www.vasculitisfoundation.org/pediatrics-vasculitis/pediatric-types/pediatric-eosinophilic-granulomatosis-with-polyangiitis/
16. Cottin V, Bel E, Bottero P, et al. Respiratory manifestations of eosinophilic granulomatosis with polyangiitis (Churg-Strauss). Eur Respir J. 2016;48(5):1429-1441. doi:10.1183/13993003.00097-2016
17. Jakes R, Kwon N, Huynh L, et al. Characterisation and burden of eosinophilic granulomatosis with polyangiitis: a European retrospective study [abstract]. Eur Respir J. 2022;60(suppl 66):2254. doi:10.1183/13993003.congress-2022.2254
18. Rice JB, White AG, Scarpati LM, et al. Long-term systemic corticosteroid exposure: a systematic literature review. Clin Ther. 2017;39(11):2216-2229. doi:10.1016/j.clinthera.2017.09.011
19. Baldini C, Talarico R, Della Rossa A, et al. Clinical manifestations and treatment of Churg-Strauss syndrome. Rheum Dis Clin North Am. 2010;36(3):527-543. doi:10.1016/j.rdc.2010.05.003
20. Scherbacher PJ, Hellmich B, Feng Y-S, et al. Prospective study of complications and sequelae of glucocorticoid therapy in ANCA-associated vasculitis. RMD Open. 2024;10(1):e003956. doi:10.1136/rmdopen-2023-003956
21. Alam V, Nanzer AM. Eosinophilic granulomatosis with polyangiitis: case report and literature review. Breathe (Sheff). 2022;18(4):220170. doi:10.1183/20734735.0170-2022
22. Hsu DC, Katelaris CH. Long-term management of patients taking immunosuppressive drugs. Aust Prescr. 2009;32(3):68-71. doi:10.18773/austprescr.2009.035